8-K
0001395937false00013959372026-02-262026-02-26
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
|
Date of Report (Date of earliest event reported): February 26, 2026 |
SYNDAX PHARMACEUTICALS, INC.
(Exact name of Registrant as Specified in Its Charter)
|
|
|
|
|
Delaware |
001-37708 |
32-0162505 |
(State or Other Jurisdiction
of Incorporation) |
(Commission File Number) |
(IRS Employer
Identification No.) |
|
|
|
|
|
730 THIRD AVENUE FLOOR 9 |
|
NEW YORK, New York |
|
10017 |
(Address of Principal Executive Offices) |
|
(Zip Code) |
|
Registrant’s Telephone Number, Including Area Code: (781) 419-1400 |
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class
|
|
Trading
Symbol(s) |
|
Name of each exchange on which registered
|
Common Stock |
|
SNDX |
|
The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02. Results of Operations and Financial Condition.
On February 26, 2026, Syndax Pharmaceuticals, Inc. (the “Company”) issued a press release and presentation announcing its financial results for the quarter and year ended December 31, 2025. A copy of the press release and presentation are furnished as Exhibits 99.1 and 99.2 to this Current Report on Form 8-K and are incorporated herein by reference.
The information contained in this Item 2.02 and in Exhibits 99.1 and 99.2 attached hereto is intended to be furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference into any of the Company’s filings under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
SYNDAX PHARMACEUTICALS, INC. |
|
|
|
|
By: |
/s/ Michael A. Metzger |
|
|
Michael A. Metzger |
|
|
Chief Executive Officer |
Dated: February 26, 2026
EX-99.1

Syndax Reports Fourth Quarter and Full Year 2025 Financial Results and Provides Business Update
•Total revenue of $68.7 million in 4Q25 and $172.4 million in FY2025 –
•Revuforj® (revumenib) net revenue of $44.2 million in 4Q25, a 38% increase vs 3Q25, and $124.8 million in FY2025 –
•Niktimvo™ (axatilimab-csfr) net revenue of $56.0 million in 4Q25, a 22% increase vs 3Q25, and $151.6 million in FY2025, resulting in Syndax collaboration revenue of $42.4 million in FY2025 –
•Completed enrollment in Phase 2 IPF trial of axatilimab; topline data expected in 4Q26 –
•Company to host a conference call today at 4:30 p.m. ET –
NEW YORK, NY, February 26, 2026 (GLOBE NEWSWIRE) – Syndax Pharmaceuticals (Nasdaq: SNDX), a commercial-stage biopharmaceutical company advancing innovative cancer therapies, today reported its financial results for the fourth quarter and full year ended December 31, 2025, and provided a business update.
“We solidified our leadership position and proved the strength of Syndax’s R&D and commercial capabilities in 2025, achieving our third FDA approval and successfully launching two first- and best-in-class medicines. We reached thousands of patients with Revuforj and Niktimvo and generated over $275 million in 2025 sales, rapidly advancing the company towards profitability,” said Michael A. Metzger, Chief Executive Officer. “With strong momentum and multiple growth drivers for both products, including increasing uptake of Revuforj in R/R NPM1m AML and the post-transplant setting, Syndax is well positioned for continued growth in 2026 and beyond.”
Mr. Metzger continued, “We’ve also made excellent progress advancing our development programs designed to further unlock multi-billion-dollar opportunities for both our medicines. We are positioned to be first to frontline AML with a menin inhibitor, and to expand our impact on chronic GVHD and other fibrotic diseases through CSF-1R inhibition. Earlier this year, we completed enrollment in our Phase 2 IPF trial and remain on track for topline data later this year which could further unlock Niktimvo’s potential as a novel antifibrotic.”
Recent Business Highlights and Anticipated Milestones
Revuforj® (revumenib)
•Achieved $44.2 million in Revuforj net revenue in the fourth quarter of 2025, a 38% increase over the third quarter of 2025. Revuforj net revenue for the full year 2025 totaled $124.8 million.
•Observed continued acceleration in demand following the FDA’s approval on October 24, 2025, of Revuforj for the treatment of relapsed or refractory (R/R) acute myeloid leukemia (AML) with a susceptible nucleophosmin 1 mutation (NPM1m) in adult and pediatric patients one year and older who have no satisfactory alternative treatment options. Total Revuforj prescriptions in the fourth quarter of 2025 were approximately 1,150, an approximate 35% increase over the third quarter of 2025.
•Initiated REVEAL-ND, a Phase 3, randomized, double-blind, placebo-controlled trial of revumenib in combination with intensive chemotherapy in newly diagnosed patients with NPM1m AML in November 2025. The trial has dual primary endpoints of measurable residual disease (MRD) negative complete remission (CR) and event free survival (EFS) to support the potential for accelerated approval and full approval, respectively.
•Received the ‘Best New Drug’ award for Revuforj at the Scrip Awards in December 2025. The award recognizes excellence in pharmaceutical development and the drug that represents the best therapeutic advance in its area.
•Highlighted the company’s leadership in menin inhibition with the presentation of 12 revumenib abstracts, including three oral presentations, at the 67th American Society of Hematology (ASH) Annual Meeting in December 2025. The presentations reported compelling results with revumenib in multiple acute leukemia subtypes across the R/R, frontline, and post-stem cell transplant maintenance setting.
•The data presented included the first real-world evidence for a menin inhibitor. The results showed an overall response rate (ORR) of 77% (10/13), a measurable residual disease (MRD) negativity rate of 75% (9/12), and favorable tolerability among primarily R/R NPM1m, KMT2Ar, and NUP98r acute leukemia patients who received revumenib in combination with standard of care therapies or as a monotherapy outside of a clinical trial.
•Multiple clinical trials evaluating revumenib across the acute leukemia treatment continuum are ongoing, such as:
•EVOLVE-2: A pivotal, Phase 3, randomized, double-blind, placebo-controlled trial of revumenib in combination with venetoclax and azacitidine in newly diagnosed NPM1m (primary efficacy analysis population) and KMT2Ar AML patients who are unfit for intensive chemotherapy. The trial is being conducted in collaboration with the HOVON network, a leading cooperative clinical trial group with extensive experience studying novel therapies for hematologic malignancies.
•REVEAL-ND: A pivotal, Phase 3, randomized, double-blind, placebo-controlled trial of revumenib in combination with intensive chemotherapy in newly diagnosed NPM1m AML patients.
•SAVE: A Phase 1/2 trial evaluating an all-oral combination of revumenib with venetoclax and decitabine/cedazuridine in pediatric and adult patients with newly diagnosed and R/R AML or mixed-lineage acute leukemia (MPAL) harboring either NPM1m, KMT2Ar, or NUP98r alterations. The trial is being conducted by investigators from MD Anderson Cancer Center. New data presented at the 2025 ASH Annual Meeting from the first cohort of newly diagnosed patients showed a complete remission (CR) rate of 76% (16/21), an ORR of 86% (18/21), and a MRD negativity rate among responders of 100% (18/18).
•Intensive chemotherapy: Two ongoing Phase 1 trials evaluating the combination of revumenib with intensive chemotherapy (7+3) in newly diagnosed NPM1m or KMT2Ar acute leukemia patients. Preliminary data presented from both trials at the 2025 ASH Annual Meeting showed that the safety profile of the combination was consistent with the profile for intensive chemotherapy alone, along with high rates of response and MRD negativity.
•BEAT AML: A Phase 1 trial evaluating the combination of revumenib with venetoclax and azacitidine in newly diagnosed older adults (≥60 years) with NPM1m or KMT2Ar AML. The trial is being conducted as part of the Leukemia & Lymphoma Society's Beat AML® Master Clinical Trial. Data presented at the 2025 European Hematology Association (EHA) Congress and simultaneously published
in the Journal of Clinical Oncology showed a CR rate of 67% (29/43), an ORR of 88% (38/43), and a MRD negativity rate of 100% (37/37) among responders.
•Post-transplant maintenance: A Phase 1 trial evaluating the safety and preliminary efficacy of revumenib as post-transplant maintenance after hematopoietic stem cell transplant (HSCT) in patients with KMT2Ar or NPM1m acute leukemia. The trial is being conducted by investigators from the City of Hope Medical Center.
•Break Through Cancer: A Phase 2 trial studying whether the combination of revumenib and venetoclax can eliminate MRD in patients with AML and extend progression-free survival. The trial is being conducted by Break Through Cancer, a collaboration between leading U.S. cancer research centers.
•INTERCEPT: A Phase 1 trial evaluating the use of novel therapies, including revumenib, to target MRD and early relapse in AML. The trial is being conducted by the Australasian Leukaemia and Lymphoma Group as part of the INTERCEPT AML master clinical trial.
•The Company expects to present additional revumenib data at major medical meetings throughout 2026, including additional real-world evidence and data from the frontline and post-HSCT maintenance setting.
•The Company expects the RAVEN trial to initiate in the second half of 2026. RAVEN is a Phase 2 collaborative trial of revumenib in combination with venetoclax and azacitidine in newly diagnosed KMT2Ar patients who would be considered eligible, or fit, for intensive chemotherapy.
Niktimvo™ (axatilimab-csfr)
•Achieved $56.0 million in Niktimvo net revenue in the fourth quarter of 2025, a 22% increase over the third quarter of 2025. Niktimvo net revenue for the full year 2025 totaled $151.6 million. Syndax and Incyte are co-commercializing Niktimvo. Syndax records 50% of the Niktimvo net commercial profit, defined as net product revenue minus the cost of sales and commercial expenses. Syndax’s share of the Niktimvo product contribution, reported as collaboration revenue, was $19.4 million and $42.4 million in the fourth quarter and full year 2025, respectively.
•Presented data from 11 axatilimab abstracts, including three oral presentations, at the 2025 ASH Annual Meeting. The abstracts highlighted the potential for axatilimab to provide long-term benefit in recurrent or refractory chronic graft-versus-host disease (GVHD) and the tolerability of axatilimab with ruxolitinib in newly diagnosed chronic GVHD.
•Presented data from nine axatilimab abstracts, including one oral presentation, at the Tandem Meetings (Transplantation & Cellular Therapy Meetings of ASTCT® and CIBMTR®) in February 2026. The data presented included a comprehensive analysis of axatilimab in patients with chronic GVHD-related bronchiolitis obliterans syndrome (BOS) in two clinical studies. The results show clinical and symptom responses across a spectrum of lung involvement.
•Two trials evaluating axatilimab in combination with standard of care therapies in newly diagnosed chronic GVHD patients are ongoing, including:
•A Phase 2, open-label, randomized, multicenter trial of axatilimab in combination with ruxolitinib in patients ≥ 12 years of age with newly diagnosed chronic GVHD.
•A pivotal Phase 3, randomized, double-blind, placebo-controlled, multi-center trial of axatilimab in combination with corticosteroids in patients ≥ 12 years of age with newly diagnosed chronic GVHD.
•Completed enrollment in MAXPIRe, a Phase 2, 26-week randomized, double-blinded, placebo-controlled trial of axatilimab on top of standard of care in patients with idiopathic pulmonary fibrosis (IPF) in the first quarter of 2026. The Company expects to report topline data in the fourth quarter of 2026.
Fourth Quarter and Full Year 2025 Financial Results
As of December 31, 2025, Syndax had cash, cash equivalents, and short-term investments of $394.1 million and 87.7 million common shares and prefunded warrants outstanding.
Total revenue for the fourth quarter of 2025 was $68.7 million, which consisted of $44.2 million in Revuforj net revenue, $19.4 million in Niktimvo collaboration revenue, and $5.1 million in milestone, license and royalty revenue. Total revenue for the full year 2025 was $172.4 million, which consisted of $124.8 million in Revuforj net revenue, $42.4 million in Niktimvo collaboration revenue, and $5.1 million in milestone, license and royalty revenue. The Niktimvo collaboration
revenue is derived from the $151.6 million in Niktimvo net revenue that was previously reported by the Company's partner Incyte for the full year 2025. Syndax records 50% of the Niktimvo net commercial profit, defined as net revenue (recorded by Incyte) minus the cost of sales and commercial expenses.
Fourth quarter 2025 research and development expenses increased to $78.6 million from $65.5 million for the comparable prior year period, and for the full year 2025 increased to $258.8 million compared to $241.6 million for 2024. The year-over-year increase was primarily due to increased clinical, medical, and employee-related expenses.
Fourth quarter 2025 selling, general and administrative expenses increased to $49.9 million from $37.7 million for the comparable prior year period, and for the full year 2025 increased to $179.7 million compared to $120.9 million for 2024. The year-over-year increase was primarily due to increased employee-related expenses and increased sales and marketing expenses related to the U.S. commercial launches of Revuforj and Niktimvo.
For the three months ended December 31, 2025, Syndax reported a net loss attributable to common stockholders of $68.0 million, or $0.78 per share, compared to a net loss attributable to common stockholders of $94.2 million, or $1.10 per share, for the comparable prior year period. For the year ended December 31, 2025, Syndax reported a net loss attributable to common stockholders of $285.4 million or $3.29 per share, compared to a net loss attributable to common stockholders of $318.8 million or $3.72 per share for the comparable prior year period.
Financial Guidance
For the full year of 2026, the Company expects total research and development plus selling, general and administrative expenses to be approximately $400 million, excluding the impact of $50 million in estimated non-cash stock compensation expense.
Syndax expects that its operating expense base will remain stable over the next couple of years. As a result, Syndax expects that its cash, cash equivalents and short-term investments, combined with its anticipated product revenue, collaboration revenue and interest income, will enable the Company to reach profitability.
Conference Call and Webcast
In connection with the earnings release, Syndax's management team will host a conference call and live audio webcast at 4:30 p.m. ET today, Thursday, February 26, 2026.
The live audio webcast and accompanying slides may be accessed through the Events & Presentations page in the Investors section of the Company's website. Alternatively, the conference call may be accessed through the following:
Conference ID: Syndax4Q25
Domestic Dial-in Number: (800) 590-8290
International Dial-in Number: (240) 690-8800
Live webcast: https://sndx-4q25.open-exchange.net
For those unable to participate in the conference call or webcast, a replay will be available on the Investors section of the Company's website at www.syndax.com approximately 24 hours after the conference call and will be available for 90 days following the call.
About Revuforj® (revumenib)
Revuforj (revumenib) is an oral, first-in-class menin inhibitor that is FDA approved for the treatment of relapsed or refractory (R/R) acute leukemia with a lysine methyltransferase 2A gene (KMT2A) translocation as determined by an FDA-authorized test in adult and pediatric patients one year and older. Revuforj is also indicated for the treatment of R/R acute myeloid leukemia (AML) with a susceptible nucleophosmin 1 (NPM1) mutation in adult and pediatric patients one year and older who have no satisfactory alternative treatment options.
Multiple trials of revumenib are ongoing or planned across the treatment landscape, including in combination with standard of care therapies in newly diagnosed patients with NPM1m or KMT2Ar AML.
Revumenib was previously granted Orphan Drug Designation for the treatment of AML, ALL and acute leukemias of ambiguous lineage (ALAL) by the U.S. FDA and for the treatment of AML by the European Commission. The U.S. FDA also granted Fast Track designation to revumenib for the treatment of adult and pediatric patients with R/R acute leukemias harboring a KMT2A rearrangement or NPM1 mutation and Breakthrough Therapy Designation for the treatment of adult and pediatric patients with R/R acute leukemia harboring a KMT2A rearrangement.
About Niktimvo™ (axatilimab-csfr)
Niktimvo (axatilimab-csfr) is a first-in-class colony stimulating factor-1 receptor (CSF-1R)-blocking antibody approved for use in the U.S. for the treatment of chronic graft-versus-host disease (GVHD) after failure of at least two prior lines of systemic therapy in adult and pediatric patients weighing at least 40 kg (88.2 lbs).
In 2016, Syndax licensed exclusive worldwide rights to develop and commercialize axatilimab from UCB. In September 2021, Syndax and Incyte entered into an exclusive worldwide co-development and co-commercialization license agreement for axatilimab in chronic GVHD and any future indications.
Axatilimab is being studied in frontline combination trials in chronic GVHD, including a Phase 2 combination trial with ruxolitinib (NCT06388564) and a Phase 3 combination trial with steroids (NCT06585774). Axatilimab is also being studied in an ongoing Phase 2 trial in patients with idiopathic pulmonary fibrosis (NCT06132256).
About Syndax
Syndax Pharmaceuticals is a commercial-stage biopharmaceutical company advancing innovative cancer therapies. Highlights of the Company's pipeline include Revuforj® (revumenib), an FDA-approved menin inhibitor, and Niktimvo™ (axatilimab-csfr), an FDA-approved monoclonal antibody that blocks the colony stimulating factor 1 (CSF-1) receptor. Fueled by our commitment to reimagining cancer care, Syndax is working to unlock the full potential of its pipeline and is conducting several clinical trials across the continuum of treatment. For more information, please visit www.syndax.com/ or follow the Company on X and LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as "anticipate," "believe," "could," "estimate," "expects," "intend," "may," "plan," "potential," "predict," "project," "should," "will," "would" or the negative or plural of those terms, and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) are intended to identify forward-looking statements. These forward-looking statements are based on Syndax's expectations and
assumptions as of the date of this press release. Each of these forward-looking statements involves risks and uncertainties. Actual results may differ materially from these forward-looking statements. Forward-looking statements contained in this press release include, but are not limited to, statements about the progress, timing, clinical development and scope of clinical trials, the reporting of clinical data for Syndax's product candidates, the acceptance of Syndax and its partners' products in the marketplace, sales, marketing, manufacturing and distribution requirements, the potential use of its product candidates to treat various cancer indications and fibrotic diseases, and Syndax's expected full year total operating expenses, including its estimated non-cash stock compensation expense. Many factors may cause differences between current expectations and actual results, including: unexpected safety or efficacy data observed during preclinical or clinical trials; clinical trial site activation or enrollment rates that are lower than expected; changes to Revuforj's or Niktimvo’s commercial availability; changes in expected or existing competition; changes in the regulatory environment; failure of Syndax's collaborators to support or advance collaborations or product candidates; and unexpected litigation or other disputes. Other factors that may cause Syndax's actual results to differ from those expressed or implied in the forward-looking statements in this press release are discussed in Syndax's filings with the U.S. Securities and Exchange Commission, including the "Risk Factors" sections contained therein. Except as required by law, Syndax assumes no obligation to update any forward-looking statements contained herein to reflect any change in expectations, even as new information becomes available.
Niktimvo is a trademark of Incyte.
All other trademarks are the property of their respective owners.
References
1. Overall response rate (ORR) includes CR, CRh, CRp, CRi, MLFS, and PR; Composite complete remission (CRc) includes CR, CRh, CRp, and CRi.
CR = Complete remission
CRh = Complete remission with partial hematologic recovery
CRp = Complete remission with incomplete platelet recovery
CRi = Complete remission with incomplete count recovery
MLFS = Morphologic leukemia-free state
PR = Partial response
Syndax Contact
Sharon Klahre
Syndax Pharmaceuticals, Inc.
sklahre@syndax.com
Tel 781.684.9827
SNDX-G
|
|
|
|
|
|
|
|
|
|
SYNDAX PHARMACEUTICALS, INC. |
|
(unaudited) |
|
CONDENSED CONSOLIDATED BALANCE SHEETS |
|
|
|
|
|
|
|
|
|
|
|
|
December 31, |
|
|
December 31, |
|
(In thousands) |
2025 |
|
|
2024 |
|
Cash, cash equivalents, short and long-term investments |
$ |
394,070 |
|
|
$ |
692,404 |
|
Total assets |
$ |
529,706 |
|
|
$ |
724,816 |
|
Total liabilities |
$ |
465,076 |
|
|
$ |
436,692 |
|
Total stockholders' equity |
$ |
64,630 |
|
|
$ |
288,124 |
|
|
|
|
|
|
|
|
|
Common stock outstanding |
|
87,405,979 |
|
|
|
85,694,443 |
|
Common stock and common stock equivalents* |
|
103,437,561 |
|
|
|
98,972,323 |
|
|
|
|
|
|
|
|
|
*Common stock and common stock equivalents: |
|
|
|
|
|
|
Common stock |
|
87,405,979 |
|
|
|
85,694,443 |
|
|
Common stock warrants (pre-funded) |
|
285,714 |
|
|
|
285,714 |
|
|
|
Common stock and pre-funded stock warrants |
|
87,691,693 |
|
|
|
85,980,157 |
|
|
Options to purchase common stock |
|
13,128,306 |
|
|
|
11,688,079 |
|
|
Restricted Stock Units |
|
2,617,562 |
|
|
|
1,304,087 |
|
|
|
Total common stock and common stock equivalents |
|
103,437,561 |
|
|
|
98,972,323 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
SYNDAX PHARMACEUTICALS, INC. |
|
(unaudited) |
|
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended December 31, |
|
|
Year Ended December 31, |
|
(In thousands, except share and per share data) |
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
Revenue |
|
|
|
|
|
|
|
|
|
|
|
|
Product revenue, net |
$ |
44,195 |
|
|
|
7,680 |
|
|
$ |
124,844 |
|
|
|
7,680 |
|
|
Collaboration revenue, net |
|
19,392 |
|
|
|
— |
|
|
|
42,367 |
|
|
|
— |
|
|
Milestone, license and royalty revenue |
|
5,141 |
|
|
|
— |
|
|
|
5,141 |
|
|
|
16,000 |
|
Total revenues |
|
68,728 |
|
|
|
7,680 |
|
|
|
172,352 |
|
|
|
23,680 |
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
Cost of product sales |
$ |
2,706 |
|
|
|
826 |
|
|
$ |
6,970 |
|
|
|
826 |
|
|
Research and development |
|
78,641 |
|
|
|
65,529 |
|
|
|
258,784 |
|
|
|
241,647 |
|
|
Selling, general and administrative |
|
49,929 |
|
|
|
37,690 |
|
|
|
179,682 |
|
|
|
120,879 |
|
Total operating expenses |
|
131,276 |
|
|
|
104,045 |
|
|
|
445,436 |
|
|
|
363,352 |
|
Loss from operations |
|
(62,548 |
) |
|
|
(96,365 |
) |
|
|
(273,084 |
) |
|
|
(339,672 |
) |
Other (expense) income, net |
|
(5,466 |
) |
|
|
2,195 |
|
|
|
(12,338 |
) |
|
|
20,914 |
|
Net loss |
$ |
(68,014 |
) |
|
$ |
(94,170 |
) |
|
$ |
(285,422 |
) |
|
$ |
(318,758 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss attributable to common stockholders |
$ |
(68,014 |
) |
|
$ |
(94,170 |
) |
|
$ |
(285,422 |
) |
|
$ |
(318,758 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per share: |
|
|
|
|
|
|
|
|
|
|
|
Basic loss per share attributable to common stockholders |
$ |
(0.78 |
) |
|
$ |
(1.10 |
) |
|
$ |
(3.29 |
) |
|
$ |
(3.72 |
) |
Diluted loss per share attributable to common stockholders |
$ |
(0.78 |
) |
|
$ |
(1.10 |
) |
|
$ |
(3.29 |
) |
|
$ |
(3.72 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted-average common shares used in calculating: |
|
|
|
|
|
|
|
|
|
|
|
Basic loss per share attributable to common stockholders |
|
87,359,323 |
|
|
|
85,791,198 |
|
|
|
86,625,610 |
|
|
|
85,622,065 |
|
Diluted loss per share attributable to common stockholders |
|
87,359,323 |
|
|
|
85,791,198 |
|
|
|
86,625,610 |
|
|
|
85,622,065 |
|

Fourth Quarter and Full Year 2025 Financial Results February 26, 2026 Exhibit 99.2

Forward-looking statements disclosure This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as "may," "will," "expect," "plan," "anticipate" and similar expressions (as well as other words or expressions referencing future events, progress, timing or circumstances) are intended to identify forward-looking statements. All statements other than statements of historical facts contained in this presentation, including statements regarding future operations, financial results and the financial condition of Syndax Pharmaceuticals, Inc. (“Syndax” or the “Company”), including financial position, strategy and plans, the progress, timing, clinical development and scope of clinical trials and the reporting of clinical data for Syndax’s product candidates, the progress of regulatory submissions and approvals and subsequent commercialization and the potential use of Syndax’s product candidates to treat various cancer indications and fibrotic diseases, and Syndax’s expectations for liquidity and future operations, are forward-looking statements. Many factors may cause differences between current expectations and actual results, including unexpected safety or efficacy data observed during preclinical studies or clinical trials, clinical site activation rates or clinical trial enrollment rates that are lower than expected; changes in expected or existing competition; the impact of macroeconomic conditions (the Russia-Ukraine war, inflation, among others) on Syndax’s business and that of the third parties on which Syndax depends, including delaying or otherwise disrupting Syndax’s clinical trials and preclinical studies, manufacturing and supply chain, or impairing employee productivity; failure of our collaborators to support or advance collaborations or product candidates and unexpected litigation or other disputes. Moreover, Syndax operates in a very competitive and rapidly changing environment. Other factors that may cause our actual results to differ from current expectations are discussed in Syndax’s filings with the U.S. Securities and Exchange Commission, including the “Risk Factors” sections contained therein. New risks emerge from time to time. It is not possible for Syndax’s management to predict all risks, nor can Syndax assess the impact of all factors on its business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statement. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this presentation may not occur and actual results could differ materially and adversely from those anticipated or implied. Except as required by law, neither Syndax nor any other person assumes responsibility for the accuracy and completeness of the forward- looking statements. Syndax undertakes no obligation to update publicly any forward-looking statements for any reason after the date of this presentation to conform these statements to actual results or to changes in Syndax’s expectations.
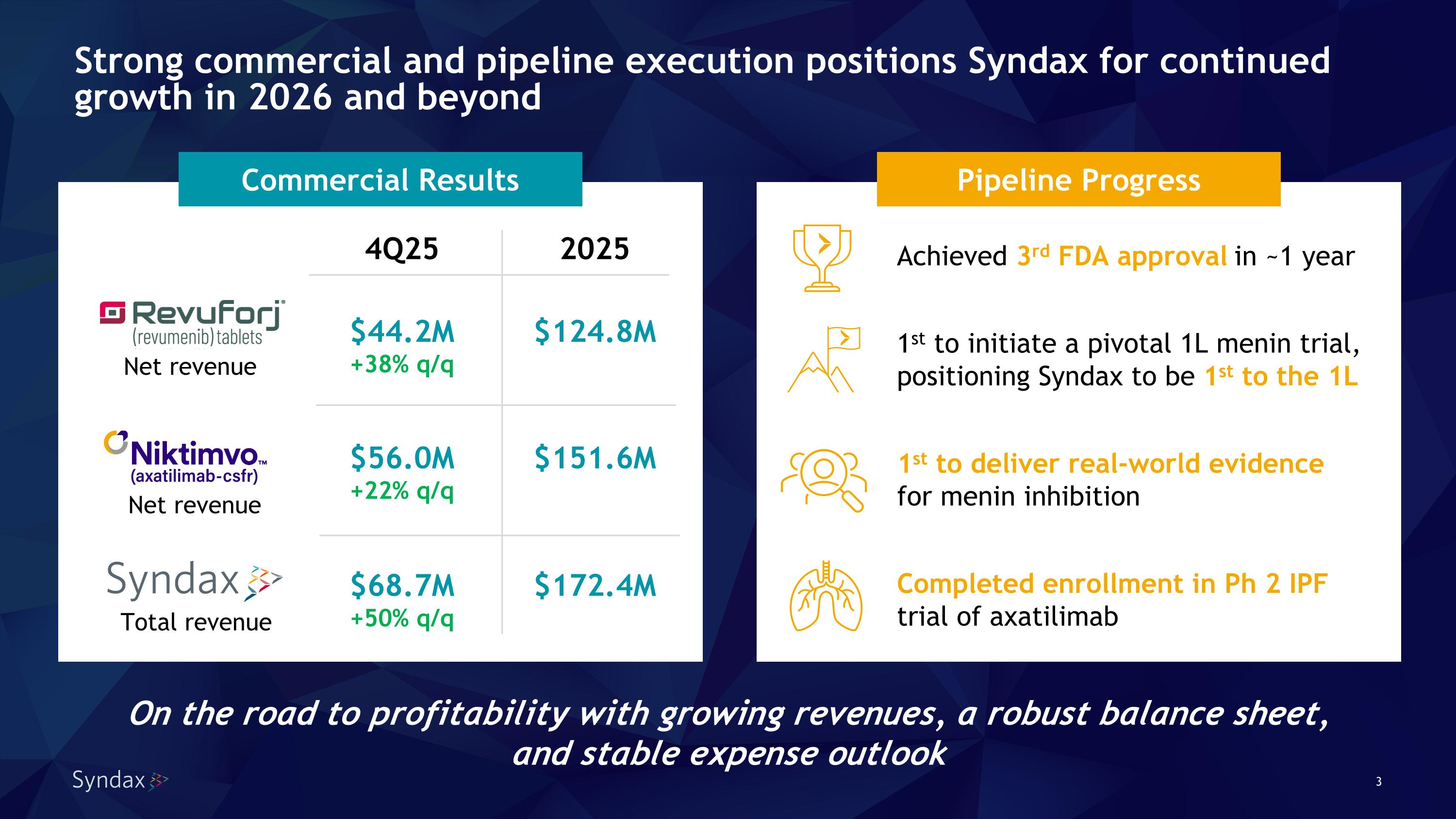
Strong commercial and pipeline execution positions Syndax for continued growth in 2026 and beyond On the road to profitability with growing revenues, a robust balance sheet, and stable expense outlook 2025 Achieved 3rd FDA approval in ~1 year 1st to initiate a pivotal 1L menin trial, positioning Syndax to be 1st to the 1L 1st to deliver real-world evidence�for menin inhibition Completed enrollment in Ph 2 IPF trial of axatilimab Pipeline Progress Commercial Results $124.8M $151.6M $172.4M 4Q25 $44.2M +38% q/q $56.0M +22% q/q $68.7M +50% q/q Total revenue Net revenue Net revenue
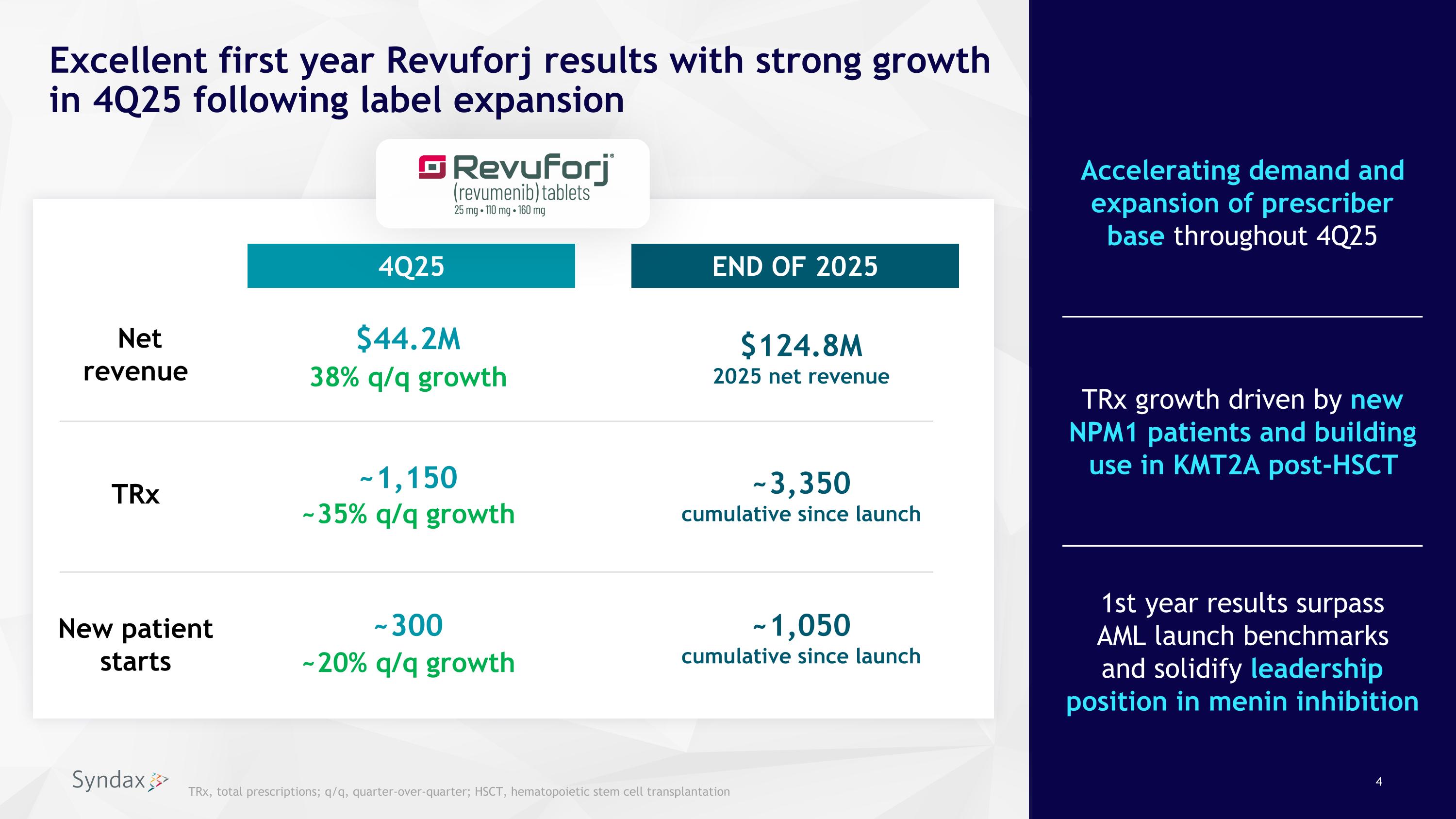
Excellent first year Revuforj results with strong growth in 4Q25 following label expansion 38% q/q growth ~20% q/q growth END OF 2025 ~3,350 cumulative since launch $44.2M 4Q25 $124.8M 2025 net revenue ~1,150 Net revenue New patient�starts ~1,050 cumulative since launch ~300 TRx ~35% q/q growth TRx, total prescriptions; q/q, quarter-over-quarter; HSCT, hematopoietic stem cell transplantation TRx growth driven by new NPM1 patients and building use in KMT2A post-HSCT 1st year results surpass�AML launch benchmarks �and solidify leadership position in menin inhibition Accelerating demand and expansion of prescriber base throughout 4Q25
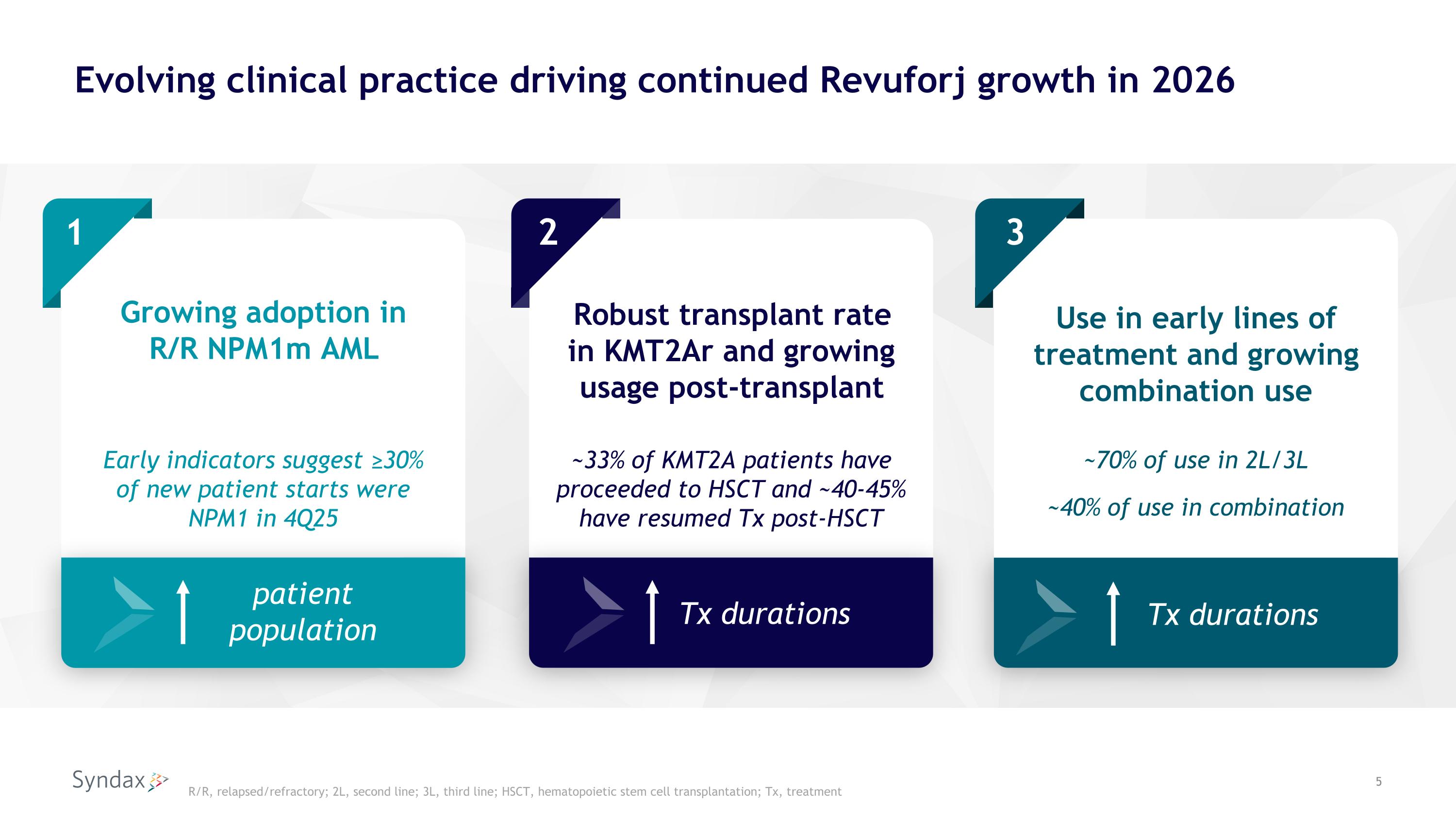
Evolving clinical practice driving continued Revuforj growth in 2026 1 2 Robust transplant rate in KMT2Ar and growing usage post-transplant Growing adoption in �R/R NPM1m AML 3 patient population Tx durations ~33% of KMT2A patients have proceeded to HSCT and ~40-45% �have resumed Tx post-HSCT Early indicators suggest ≥30% of new patient starts were NPM1 in 4Q25 R/R, relapsed/refractory; 2L, second line; 3L, third line; HSCT, hematopoietic stem cell transplantation; Tx, treatment Use in early lines of�treatment and growing combination use Tx durations ~70% of use in 2L/3L ~40% of use in combination
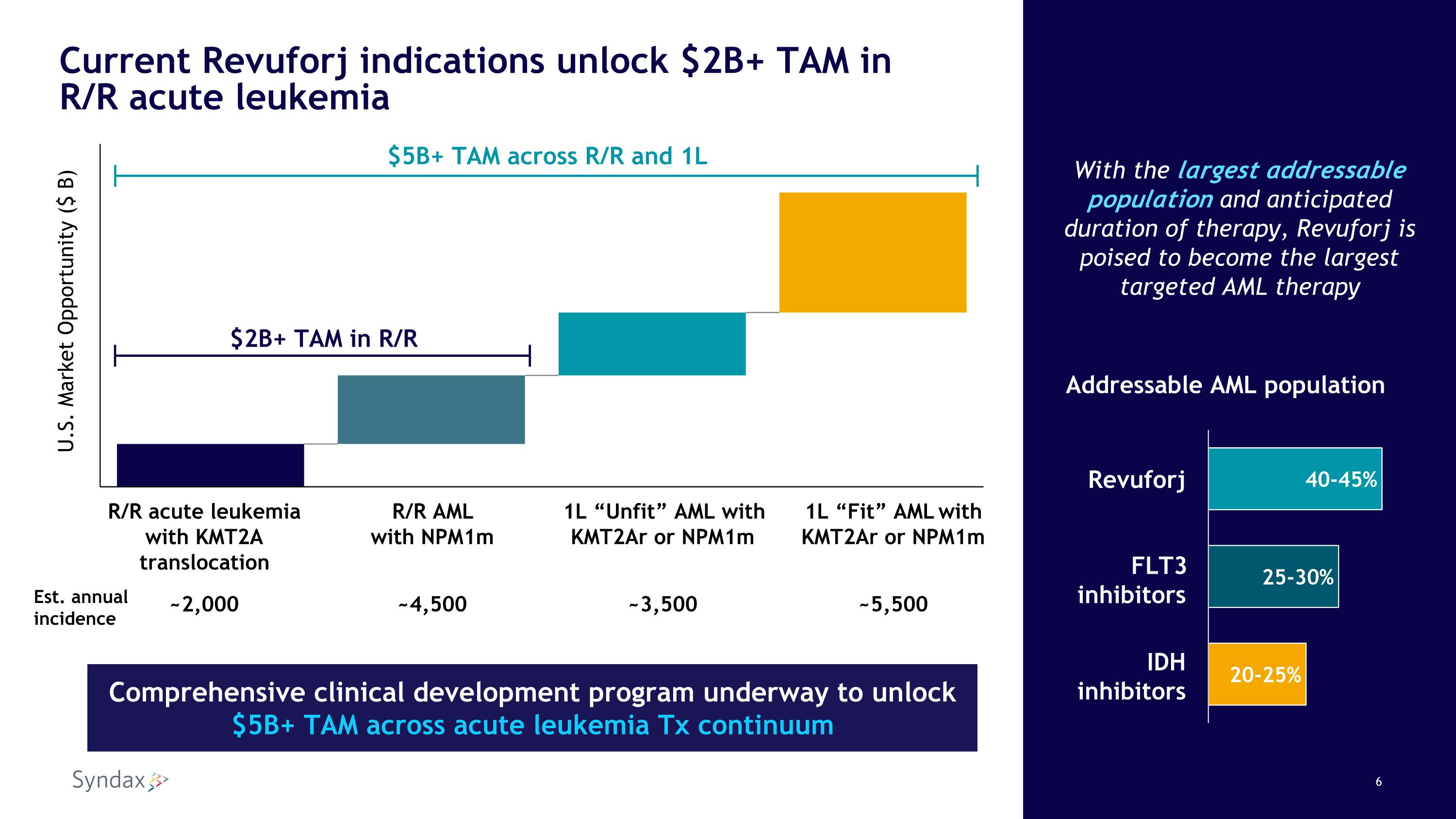
Current Revuforj indications unlock $2B+ TAM in�R/R acute leukemia With the largest addressable population and anticipated duration of therapy, Revuforj is poised to become the largest targeted AML therapy R/R acute leukemia with KMT2A translocation R/R AML �with NPM1m 1L “Unfit” AML with KMT2Ar or NPM1m 1L “Fit” AML with KMT2Ar or NPM1m ~2,000 ~4,500 ~3,500 ~5,500 U.S. Market Opportunity ($ B) $5B+ TAM across R/R and 1L $2B+ TAM in R/R Addressable AML population Revuforj FLT3 inhibitors IDH inhibitors Comprehensive clinical development program underway to unlock �$5B+ TAM across acute leukemia Tx continuum Est. annual�incidence
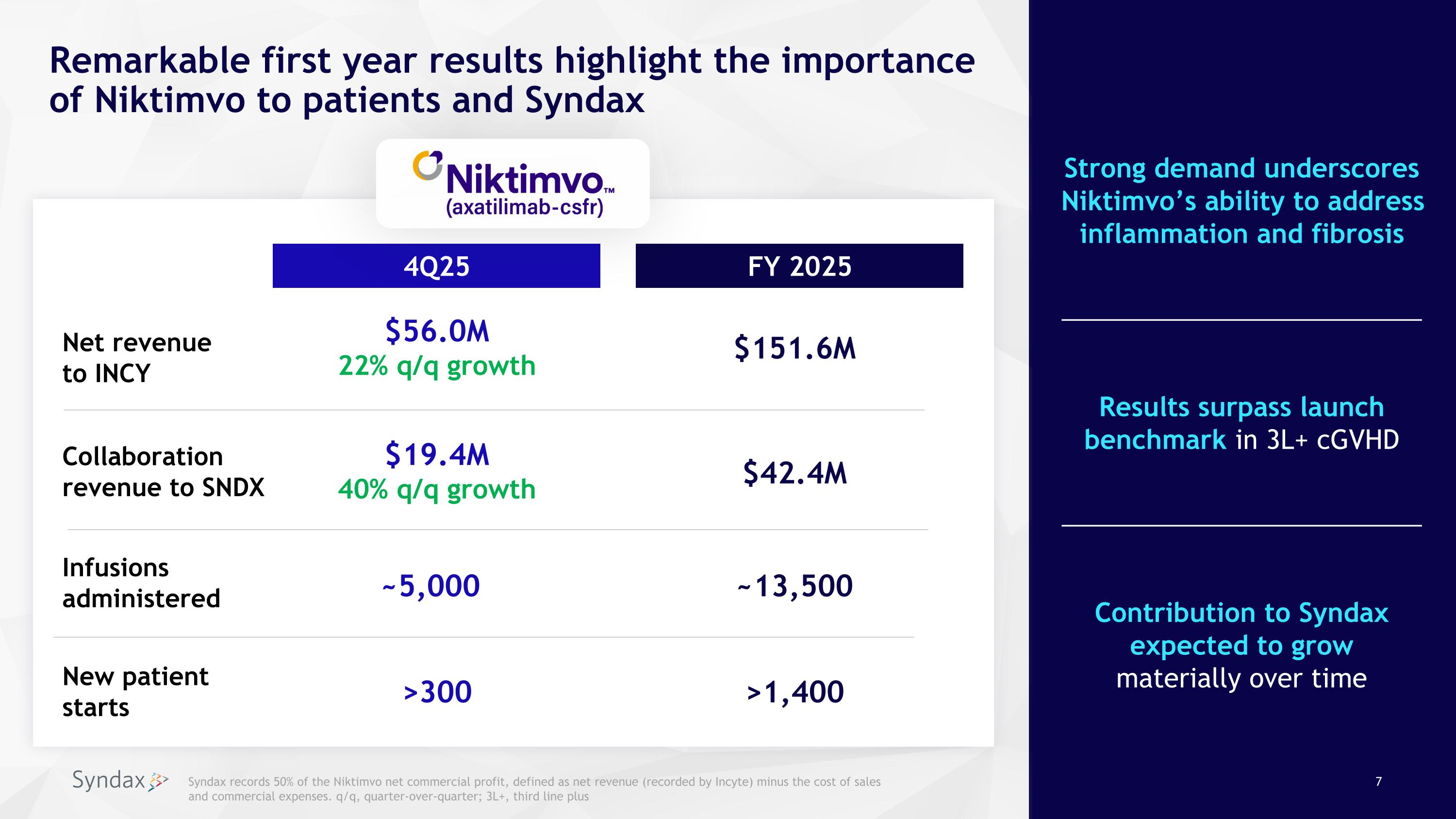
Remarkable first year results highlight the importance of Niktimvo to patients and Syndax FY 2025 ~13,500 $56.0M 22% q/q growth 4Q25 $151.6M ~5,000 Net revenue to INCY New patient�starts >1,400 >300 Infusions administered Collaboration revenue to SNDX $19.4M 40% q/q growth $42.4M Syndax records 50% of the Niktimvo net commercial profit, defined as net revenue (recorded by Incyte) minus the cost of sales�and commercial expenses. q/q, quarter-over-quarter; 3L+, third line plus Contribution to Syndax expected to grow materially over time Results surpass launch benchmark in 3L+ cGVHD Strong demand underscores Niktimvo’s ability to address inflammation and fibrosis
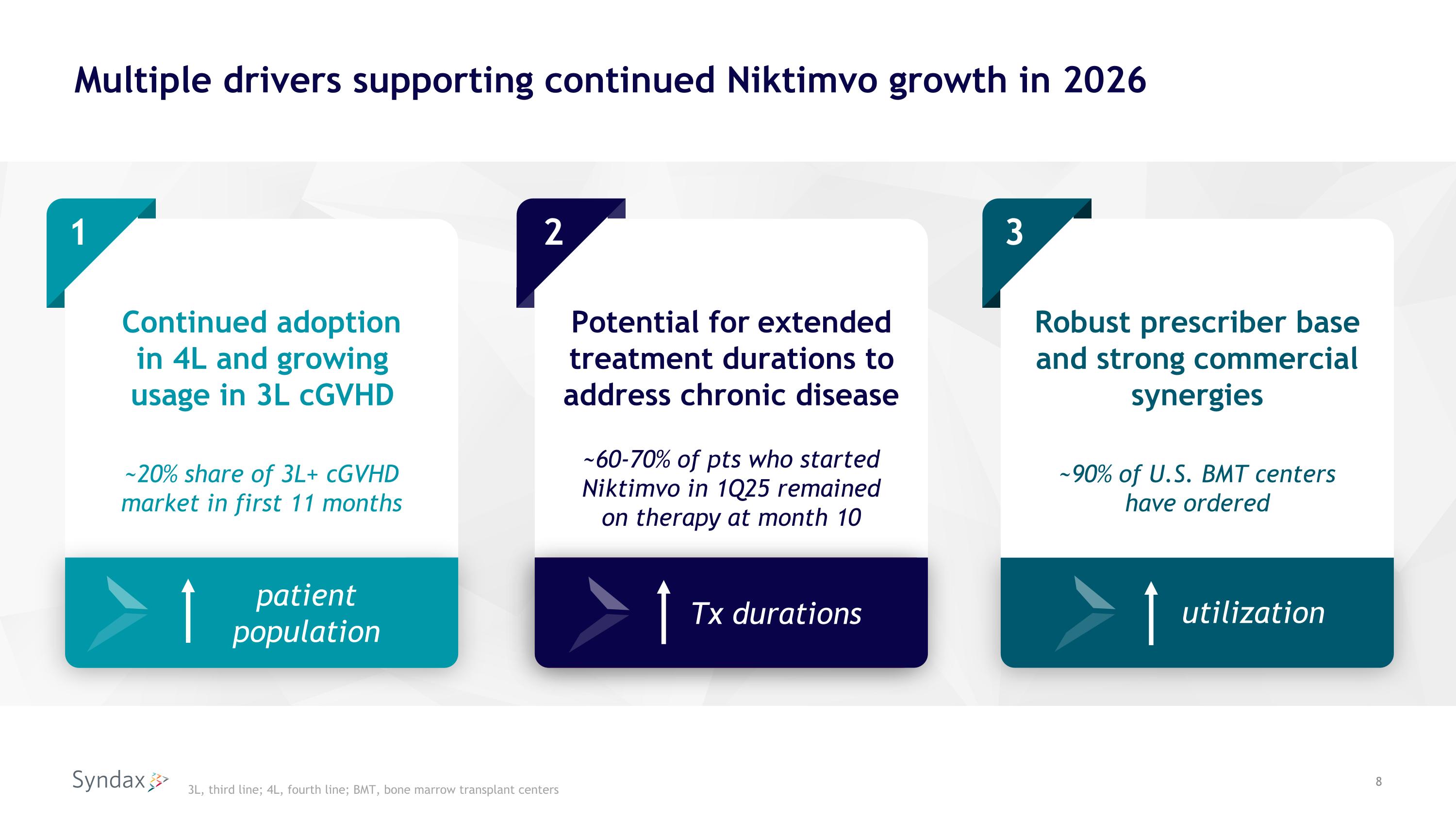
Multiple drivers supporting continued Niktimvo growth in 2026 1 Continued adoption in 4L and growing usage in 3L cGVHD 2 Potential for extended treatment durations to address chronic disease Robust prescriber base and strong commercial synergies 3 3L, third line; 4L, fourth line; BMT, bone marrow transplant centers ~20% share of 3L+ cGVHD market in first 11 months ~60-70% of pts who started Niktimvo in 1Q25 remained on therapy at month 10 Tx durations patient population ~90% of U.S. BMT centers have ordered utilization 3
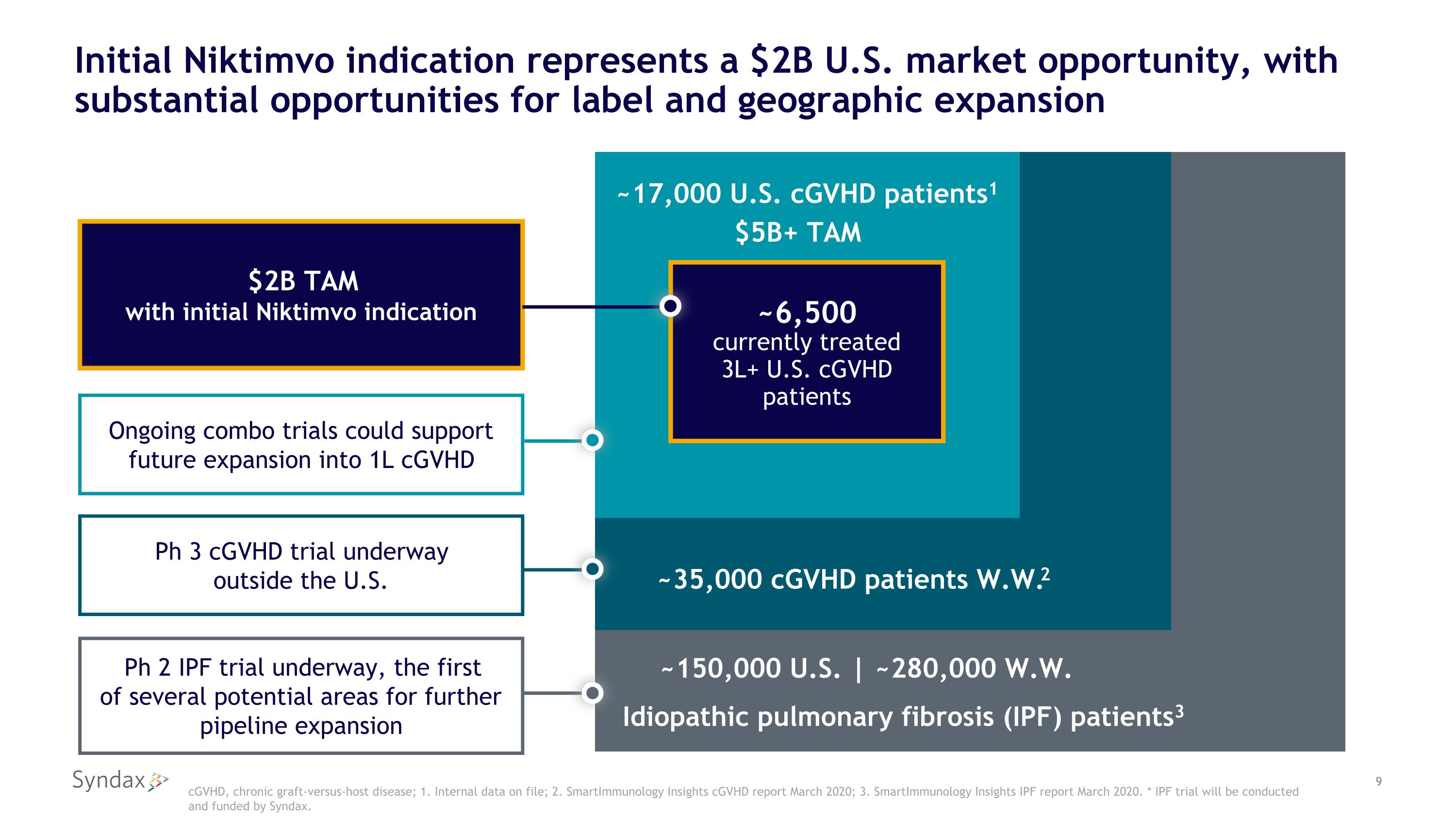
Initial Niktimvo indication represents a $2B U.S. market opportunity, with substantial opportunities for label and geographic expansion cGVHD, chronic graft-versus-host disease; 1. Internal data on file; 2. SmartImmunology Insights cGVHD report March 2020; 3. SmartImmunology Insights IPF report March 2020. * IPF trial will be conducted and funded by Syndax. ~17,000 U.S. cGVHD patients1 ~6,500 currently treated 3L+ U.S. cGVHD patients ~35,000 cGVHD patients W.W.2 Idiopathic pulmonary fibrosis (IPF) patients3 ~150,000 U.S. | ~280,000 W.W. $2B TAM �with initial Niktimvo indication Ongoing combo trials could support future expansion into 1L cGVHD Ph 2 IPF trial underway, the first�of several potential areas for further pipeline expansion Ph 3 cGVHD trial underway�outside the U.S. $5B+ TAM

Revumenib (select trials) Ph 1 Ph 2 Ph 3 FDA �Approved Setting Study Name Regimen NPM1m KMT2Ar NUP98r R/R AUGMENT-101 Rev mono ● ● AUGMENT-102 Rev + IC ● ● ● SAVE Rev + ven/oral HMA ● ● ● Post-HSCT Maintenance Ball study Rev mono ● ● 1L Unfit�for IC BEAT AML Rev + ven/aza ● ● SAVE Rev + ven/oral HMA ● ● ● EVOLVE-2 Rev + ven/aza ● ● Fit�for IC 708 and NCI Rev + IC ● ● ● REVEAL-ND Rev + IC ● RAVEN Rev + ven/aza ● Axatilimab (select trials) Ph 1 Ph 2 Ph 3 FDA Approved Setting Study Name Regimen R/R cGVHD AGAVE-201 Axa mono 1L cGVHD AXemplify-357* Axa + corticosteroids NCT06388564* Axa + ruxolitinib IPF MAXPIRe Axa on top of SOC Laser-focused on unlocking the full potential of menin and CSF-1R inhibition Rev, revumenib; Axa, axatilimab; IC, intensive chemotherapy; 1L, frontline; R/R, relapsed or refractory; ven/aza, venetoclax and azacitidine; HMA, hypomethylating agent; HSCT, hematopoietic�stem cell transplantation. *Trials led by Incyte. List is not inclusive of ongoing ex-U.S. trials. Global enrollment underway in pivotal 1L trials with dual primary endpoints to support potential for accelerated and full approval 1L, maintenance, & real-world data anticipated throughout 2026 at major medical meetings Two ongoing 1L cGVHD trials; topline axa + rux data anticipated early 2027 and axa + steroids early 2028 Completed enrollment in MAXPIRe; topline data anticipated in 4Q26
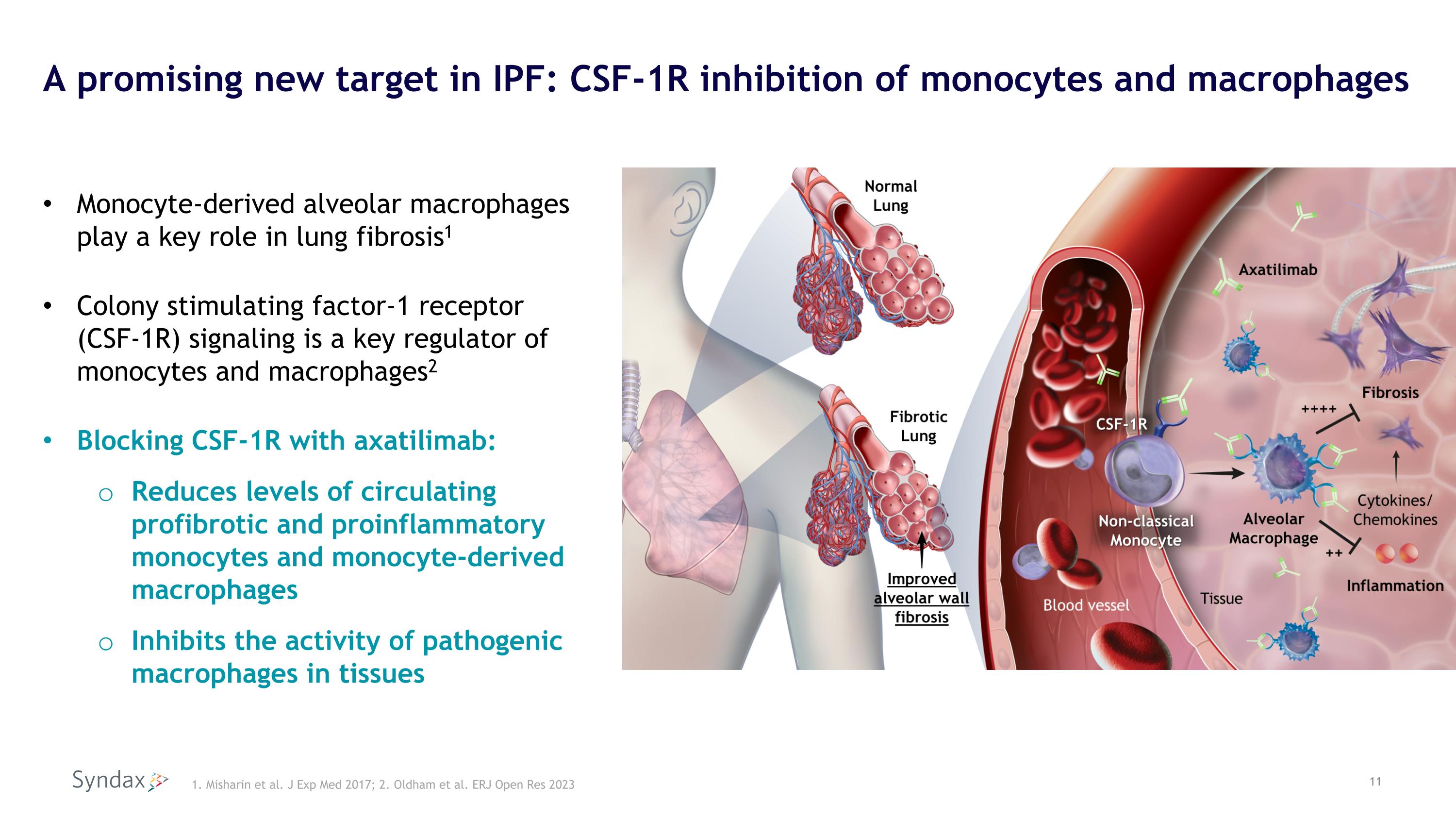
A promising new target in IPF: CSF-1R inhibition of monocytes and macrophages Monocyte-derived alveolar macrophages play a key role in lung fibrosis1 Colony stimulating factor-1 receptor (CSF-1R) signaling is a key regulator of monocytes and macrophages2 Blocking CSF-1R with axatilimab: Reduces levels of circulating profibrotic and proinflammatory monocytes and monocyte-derived macrophages Inhibits the activity of pathogenic macrophages in tissues 1. Misharin et al. J Exp Med 2017; 2. Oldham et al. ERJ Open Res 2023 11
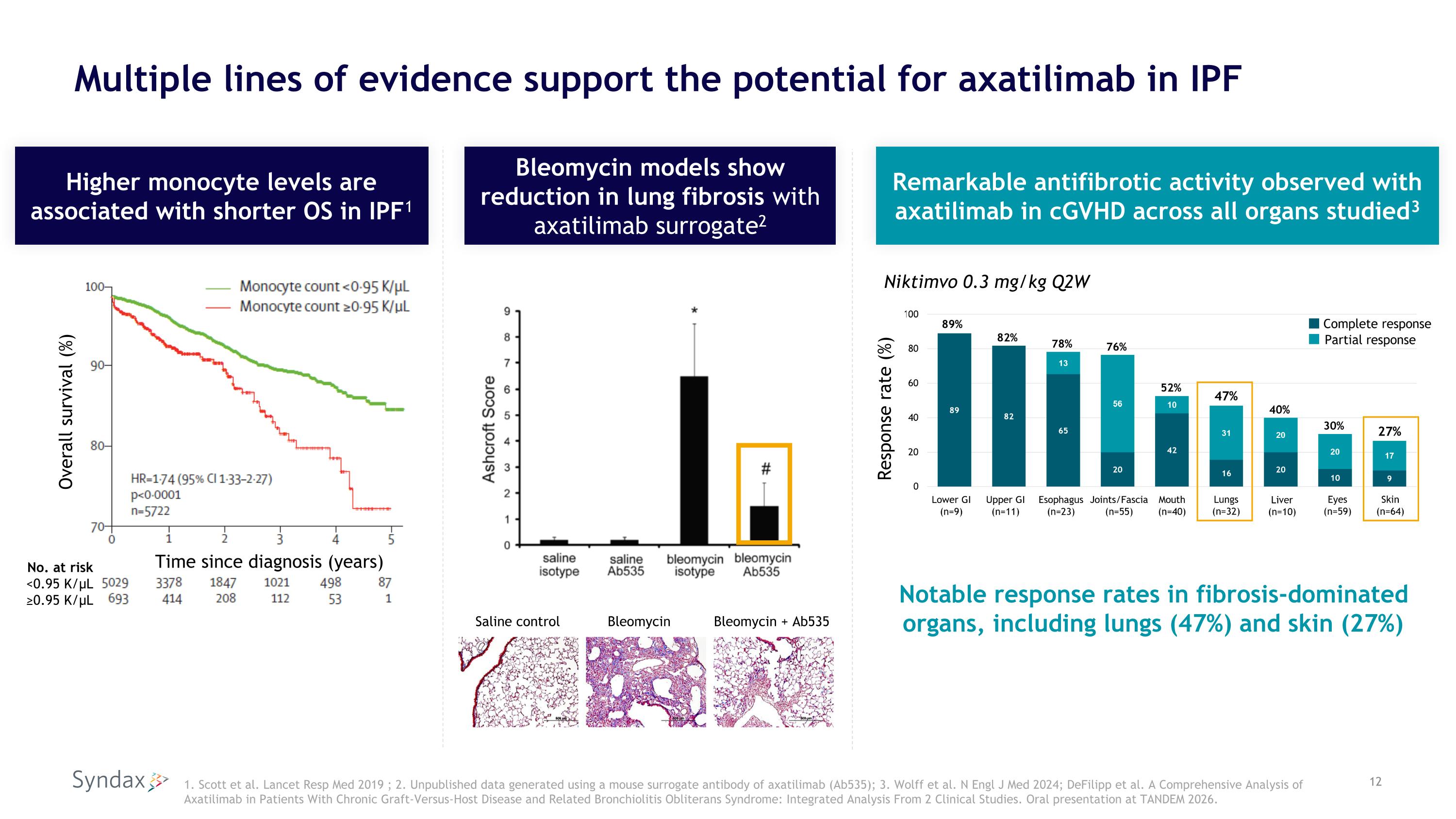
Multiple lines of evidence support the potential for axatilimab in IPF 1. Scott et al. Lancet Resp Med 2019 ; 2. Unpublished data generated using a mouse surrogate antibody of axatilimab (Ab535); 3. Wolff et al. N Engl J Med 2024; DeFilipp et al. A Comprehensive Analysis of Axatilimab in Patients With Chronic Graft-Versus-Host Disease and Related Bronchiolitis Obliterans Syndrome: Integrated Analysis From 2 Clinical Studies. Oral presentation at TANDEM 2026. Overall survival (%) No. at risk <0.95 K/µL ≥0.95 K/µL Time since diagnosis (years) Higher monocyte levels are associated with shorter OS in IPF1 Remarkable antifibrotic activity observed with axatilimab in cGVHD across all organs studied3 Bleomycin models show reduction in lung fibrosis with axatilimab surrogate2 Saline control Bleomycin Bleomycin + Ab535 Niktimvo 0.3 mg/kg Q2W Notable response rates in fibrosis-dominated organs, including lungs (47%) and skin (27%) Response rate (%)
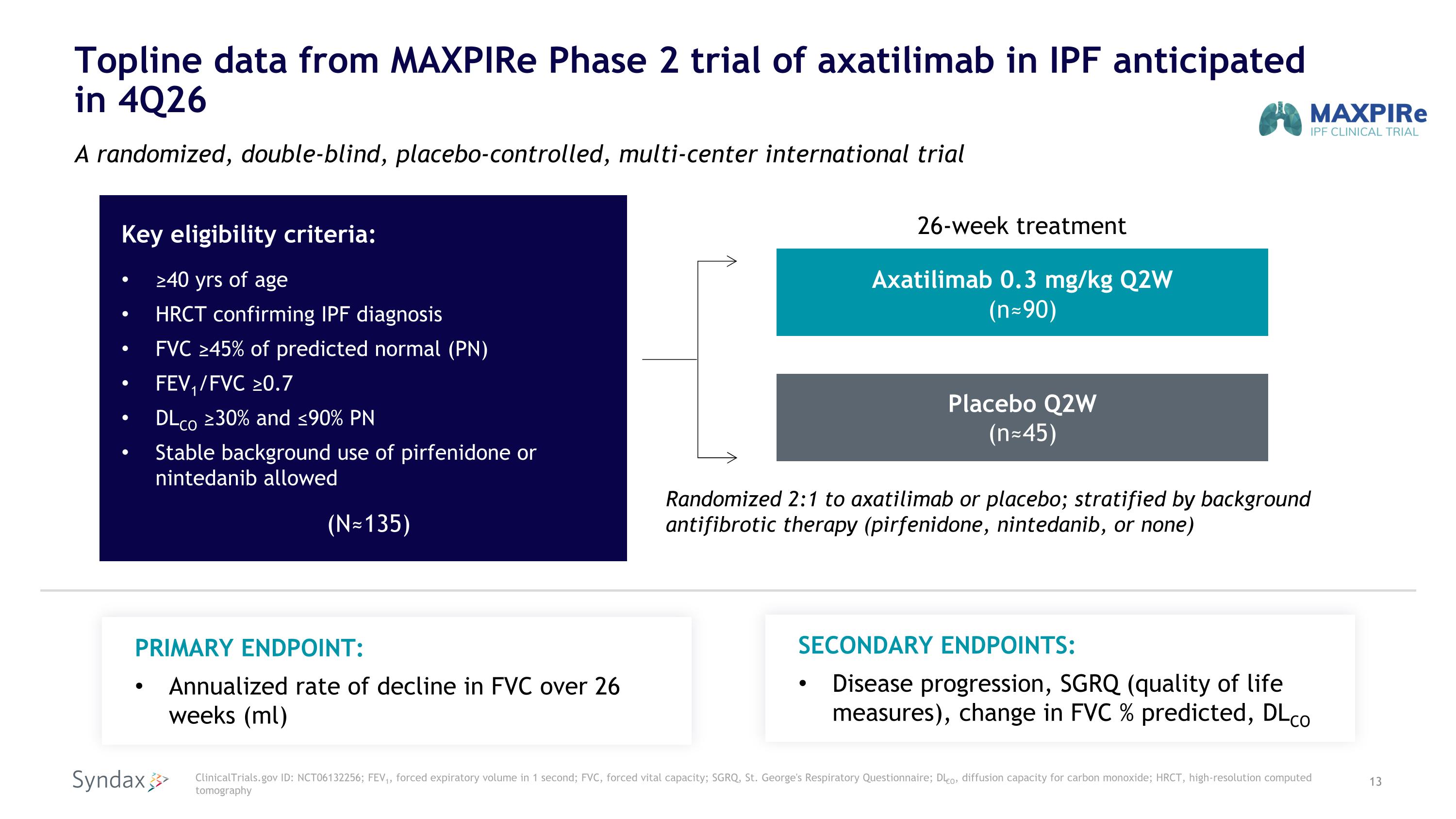
Topline data from MAXPIRe Phase 2 trial of axatilimab in IPF anticipated�in 4Q26 A randomized, double-blind, placebo-controlled, multi-center international trial Key eligibility criteria: ≥40 yrs of age HRCT confirming IPF diagnosis FVC ≥45% of predicted normal (PN) FEV1/FVC ≥0.7 DLCO ≥30% and ≤90% PN Stable background use of pirfenidone or nintedanib allowed (N≈135) Placebo Q2W�(n≈45) ClinicalTrials.gov ID: NCT06132256; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; SGRQ, St. George's Respiratory Questionnaire; DLCO, diffusion capacity for carbon monoxide; HRCT, high-resolution computed tomography PRIMARY ENDPOINT: Annualized rate of decline in FVC over 26 weeks (ml) Axatilimab 0.3 mg/kg Q2W �(n≈90) Randomized 2:1 to axatilimab or placebo; stratified by background antifibrotic therapy (pirfenidone, nintedanib, or none) SECONDARY ENDPOINTS: Disease progression, SGRQ (quality of life measures), change in FVC % predicted, DLCO 26-week treatment
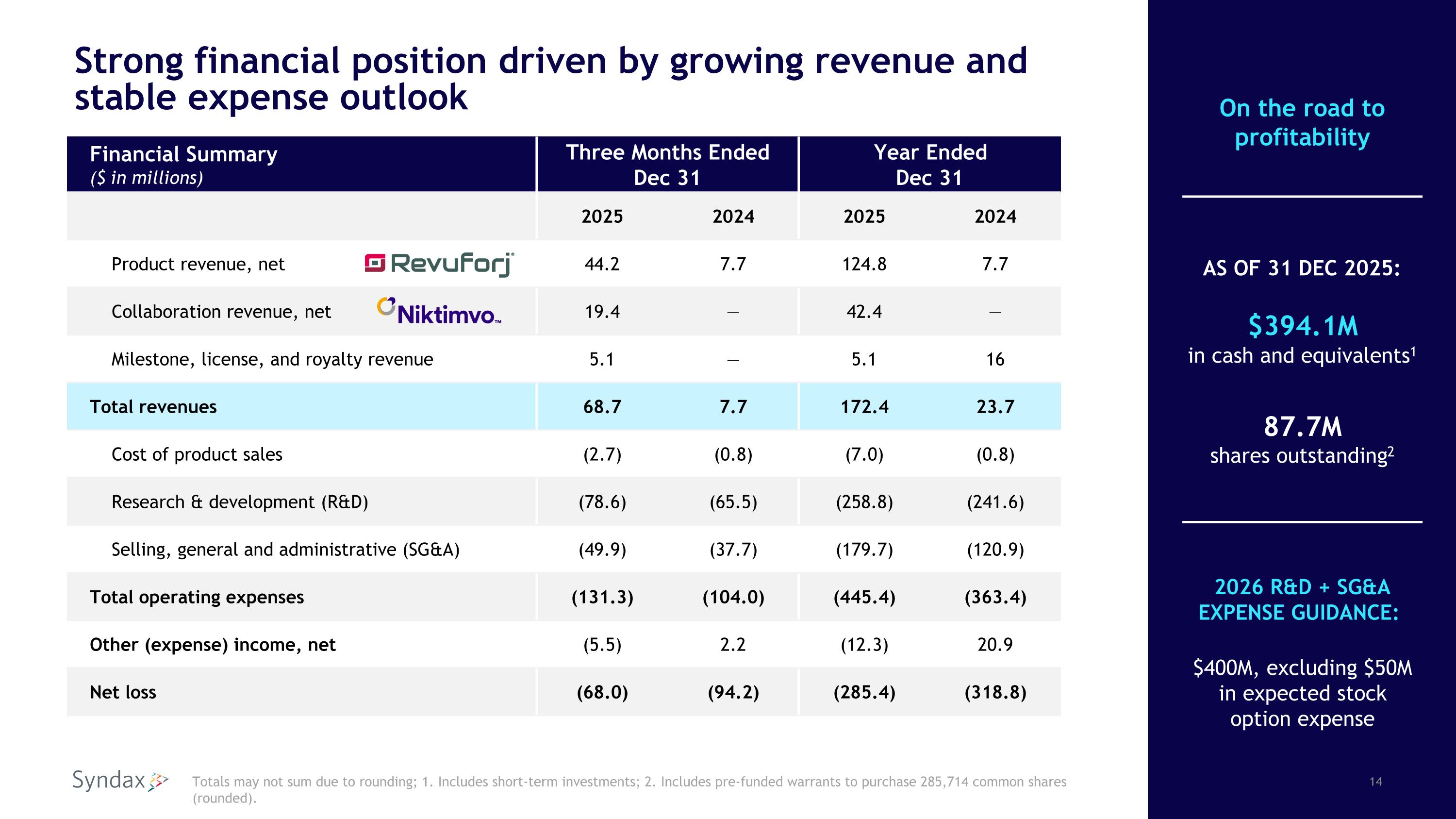
Strong financial position driven by growing revenue and stable expense outlook Financial Summary ($ in millions) Three Months Ended�Dec 31 Year Ended Dec 31 2025 2024 2025 2024 Product revenue, net 44.2 7.7 124.8 7.7 Collaboration revenue, net 19.4 — 42.4 — Milestone, license, and royalty revenue 5.1 — 5.1 16 Total revenues 68.7 7.7 172.4 23.7 Cost of product sales (2.7) (0.8) (7.0) (0.8) Research & development (R&D) (78.6) (65.5) (258.8) (241.6) Selling, general and administrative (SG&A) (49.9) (37.7) (179.7) (120.9) Total operating expenses (131.3) (104.0) (445.4) (363.4) Other (expense) income, net (5.5) 2.2 (12.3) 20.9 Net loss (68.0) (94.2) (285.4) (318.8) Totals may not sum due to rounding; 1. Includes short-term investments; 2. Includes pre-funded warrants to purchase 285,714 common shares (rounded). AS OF 31 DEC 2025: $394.1M �in cash and equivalents1 87.7M�shares outstanding2 2026 R&D + SG&A EXPENSE GUIDANCE: $400M, excluding $50M in expected stock option expense On the road to profitability

Proven execution supports continued focus on revenue growth, pipeline progress, and data generation 2026 Anticipated Milestones 2025 Key Accomplishments Advance global enrollment in pivotal 1L trials of revumenib Publish & present industry leading clinical data, including 1L, maintenance, and real-world evidence for revumenib Report topline Ph 2 axatilimab data in IPF in 4Q26 Initiate RAVEN 1L trial of revumenib in fit KMT2Ar in 2H26 Initiate a program to generate proof-of-principle clinical �data with revumenib in myelofibrosis Executed two strong product launches Expanded Revuforj into 2nd indication Initiated 1st pivotal 1L trial of a menin inhibitor Presented first RWE for a menin inhibitor Initiated managed access program, expanding access to Revuforj in certain OUS regions 1L, frontline; RWE, real-world evidence; OUS, outside the United States


















